Spirally splitting sporangia
- Aglaophyton
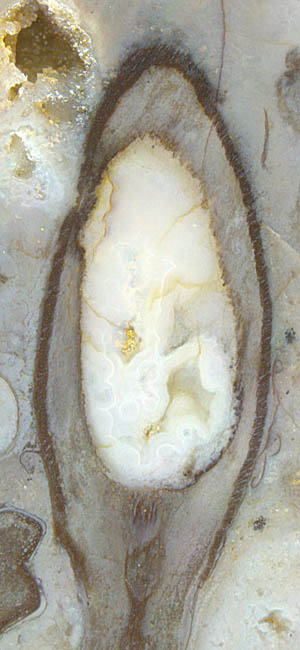 As a well known fact, the sporangia of Aglaophyton,
a common plant in the Lower Devonian Rhynie chert, are borne
upright at the end of shoots.
Although their shape is axially symmetric, the cellular pattern of the
outer wall is not. This is seen in the lengthwise section
in Fig.1,
where a close look at the wall reveals that all cells, where visible,
are leaning to the right, indicating a twist of the whole pattern.
As a well known fact, the sporangia of Aglaophyton,
a common plant in the Lower Devonian Rhynie chert, are borne
upright at the end of shoots.
Although their shape is axially symmetric, the cellular pattern of the
outer wall is not. This is seen in the lengthwise section
in Fig.1,
where a close look at the wall reveals that all cells, where visible,
are leaning to the right, indicating a twist of the whole pattern.
Splitting of the capsule (Fig.2) is an easy and
apparently adequate way of shedding the spores. It is not obvious why
the more
advanced contemporaneous plants had developed special means of spore
release, as
holes at the top of every compartment of the branched capsule (Horneophyton),
or valves opening along pre-formed weak lines in the capsule walls.
(Nothia,
Trichopherophyton, Ventarura, Asteroxylon).
One may wonder why the capsule in Fig.1 is empty but not split.
Either the fissure is small and not seen on this section or there is
none and the spores were completely consumed by some spore eater going
in and out through a hole gnawed into the wall above left. (Although
sporangia with a hole in the wall are not rare, they were not known in
the scientific literature and have been
described first in Rhynie
Chert News 7.)
Fig.1 (far right): Aglaophyton
sporangium, empty, with the cells of
the dark outer wall in a lopsided array.
Width of the capsule 4.7mm.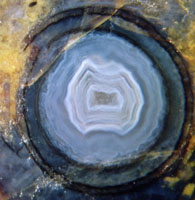
Fig.2: Cross-section of split
and empty Aglaophyton
sporangium
Width
of the capsule 3.2 mm.
The
split and apparently twisted capsules had led to the idea that the
twist came with
the splitting [1,2], similar as with numerous seed plants whose pods
become
mechanically stressed in maturation, then release the stored elastic
energy while bursting and scattering the seeds
with a jerk. No contrary
evidence had been available, judging from the statement in [2] that
"... the
alignment of the epidermal and other wall layers in intact sporangia is
not known."
The
alignment of the epidermal wall layer in non-split sporangia is known
now.
Evidence is
provided by the rare cases of sporangia seen from outside, as
in Rhynie
Chert News 5
and
in Figs.3,4 below. The latter show a slight growth anomaly which
incidentally
offers a unique view on the spiral texture of the cell array. Unawares
of the sporangium and its shape and orientation, the cut plane has been
chosen such that it nearly coincides with the tangential plane of a
saddle
point of the sporangium curved like a
cucumber. (Note that the normal
sporangium,
spindle-shaped as in Fig.1 or more cylindrical, does not have saddle
points, which are characterized by concave curvature in one direction
and convex curvature in another one.)
The sporangium is not split open but the prospective
splitting paths are clearly seen, arranged not at random but with
roughly equal spacing. Apparently they do not
or not always extend over the length of the capsule. One
may wonder why they look conspicuously straight although they are
supposed to be part of 3D-spirals or screw lines. The straight
aspect is due to an interesting mathematical fact: Every screw line, or
every turn of a thread, if looked at straight on, is seen as a curve
which changes its sign of apparent curvature at the point nearest to
the observer. This means the apparent curvature is zero there and
nearly
zero for some distance on either side, and if the more curved ends are
hidden in the depth as in the below pictures, the visible parts of the
screw lines appear straight.


Figs.3,4
(left): Aglaophyton
sporangium misshapen as a cucumber, offering a
rare view on the wall texture with prospective splitting paths.
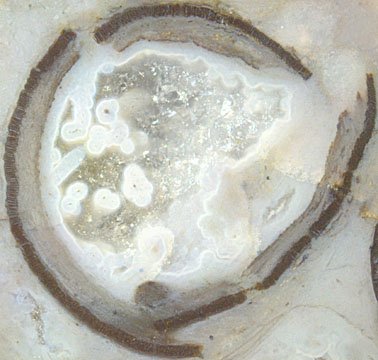
Fig.5: Aglaophyton
sporangium, about 7mm across before splitting.
Not closely related to the present problem
of spiral splitting but nevertheless interesting are the
following facts:
An uncommonly big sporangium (Fig.5) with
estimated original diameter of 7mm is incompatible with the
(slightly contradictory) size data in [2]: "Sporangia ... < 12mm
long and < 4mm wide, ..." and, on the next page, "... up to 12mm
long and 5mm wide, ...". Wider sporangia are
not quite rare: see Rhynie
Chert News 11. The
separation of the
capsule parts in Fig.5 may be due to later deformation.
The
inner cavity of hollow sporangia, like that of hollow shoots, often
looks distinctly different from the surrounding chert matrix. Even
split sporangia show such difference indicating a different sequence
of silicification
stages: See the blue agate in Fig.2 and coarse quartz
crystals in Figs.3,4,5. No
satisfactory explanation can be proposed here at present but one
observation on Fig.5 may eventually give a clue: Tiny dark dots in
white circles are cross-sections of aquatic fungus
hyphae which throve in the water-filled cavity
before they became coated
with silica gel turned into whitish chalcedony, and the remaining space
eventually became filled with coarse quartz. The same has repeatedly
been observed with former gas bubbles which had formed and got
stuck in the waterlogged habitat, got fixed by silicification of the
water, later
became water-filled as the gas gradually escaped by diffusion through
the silica gel while water and dissolved silica
entered likewise. Hence, similar processes might have been going on
within empty sporangia with hyphae seen inside.
No gap is seen in Fig.2 despite of the split. Certainly the dry
capsule had a gap for spore release which
possibly closed after inundation.
As another remarkabe fact, the thread of the cell pattern is always
right-handed. It would be
interesting to know whether or not there is a deep relation to the
right-handedness of other objects: sporangia
of other plant species, charophyte whorls, and
the enigmatic Nematoplexus,
for example.
Samples: Figs.1,5: Rh6/38.2, Fig.2: Rh20/2.2, Figs.3,4: Rh7/31.1,
Despite of some remaining uncertainty in the interpretation of Aglaophyton
fossils, the
observations reported here seem to justify the following assumptions:
(1) The spiral texture of the sporangium wall had
been there before splitting.
(2) The position of the split in the wall is not pre-determined.
(3) There are several prospective split lines, spaced apart by about
20-30 cell files.
(4) Not every prospective split line runs along the whole sporangium.
(5) Which one of the prospective split lines becomes active is left to
chance.
H.-J.
Weiss
2014
[1] W.
Remy: Der Dehiszenzmechanismus
der Sporangien von Rhynia*,
Argumenta Palaeobotanica 5(1978),
23-30. * re-named Aglaophyton in [2]
[2] David
S. Edwards, Aglaophyton
major, a non-vascular** land-plant from the Devonian
Rhynie Chert,
Bot. J. Linn. Soc. 93(1986),
173-204.
** doubted in [3]
[3] Dianne
Edwards : A review of the sporophytes of embryophytes
in the cherts at Rhynie,
Trans. Roy. Soc. Edinburgh, Earth
Sciences 94(2004 for 2003), 397-410.
 |
 |
62 |


 As a well known fact, the sporangia of Aglaophyton,
a common plant in the Lower Devonian Rhynie chert, are borne
upright at the end of shoots.
Although their shape is axially symmetric, the cellular pattern of the
outer wall is not. This is seen in the lengthwise section
in Fig.1,
where a close look at the wall reveals that all cells, where visible,
are leaning to the right, indicating a twist of the whole pattern.
As a well known fact, the sporangia of Aglaophyton,
a common plant in the Lower Devonian Rhynie chert, are borne
upright at the end of shoots.
Although their shape is axially symmetric, the cellular pattern of the
outer wall is not. This is seen in the lengthwise section
in Fig.1,
where a close look at the wall reveals that all cells, where visible,
are leaning to the right, indicating a twist of the whole pattern.




